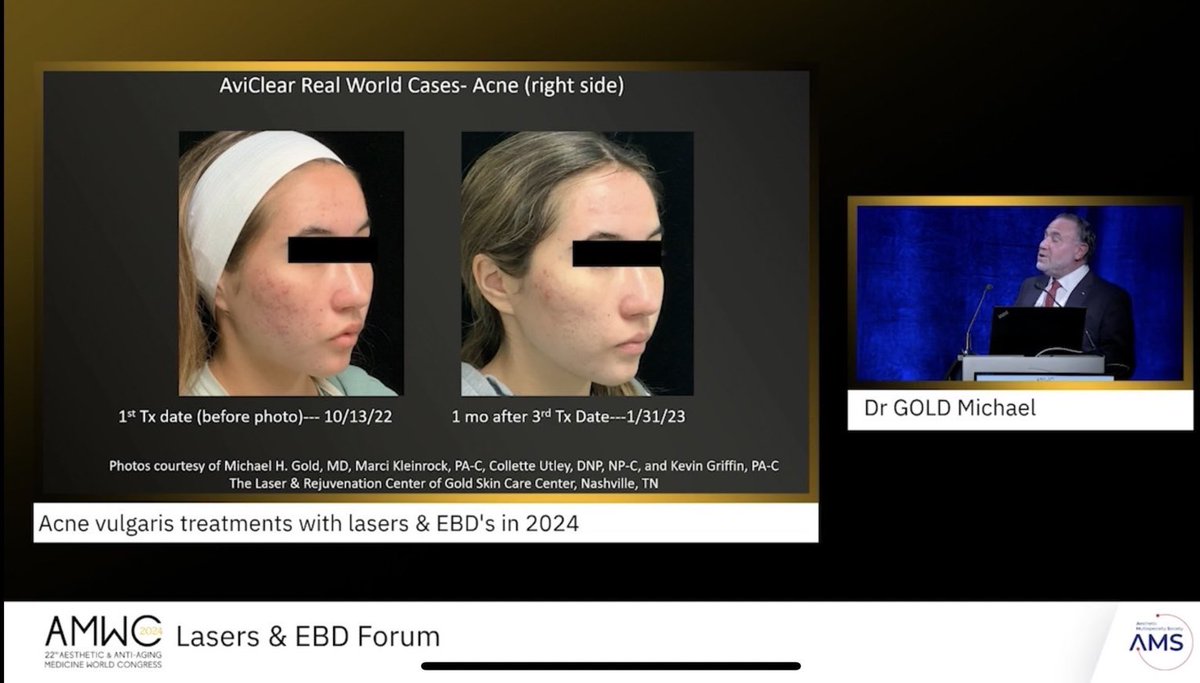
USFDA rejects another China 🇨🇳 Made IO inspite of wide separation of OS curves . Camrelizumb plus Rivoceranib combination for Advanced HCC is rejected by U.S. FDA . Free HOME Run for Atezolizumab plus bev ? 🤔 Tanios Bekaii-Saab, MD Axel Grothey


Clarification by #LaurusLabs on this USFDA news going on in market as that co contributes only 1% of the Consolidated Revenue.

#LAURUSLABS | 📌 Laurus Labs Limited clarified the USFDA inspection at its subsidiary, LSPL, and the untitled letter. LSPL is not a material subsidiary, contributes less than 1% of consolidated revenues, and has not filed any US DMFs.
Details: sqst.in/ysn1p
⚡ AI

आंखों की पुतली को फैलाने के लिए #CaplinPoint arm को Abbreviated New Drug Application (ANDA) Phenylephrine Hydrochloride Ophthalmic के लिए #USFDA मंज़ूरी मिली।
#stock #sharemarket #business #news #marketupdates #trending


#LaurusLabs CEO, Satyanarayana Chava is confident of strong growth in CDMO business this year
Here's more from the interview with Sonia Shenoy , Prashant Nair and Nigel D'Souza 👇
cnbctv18.com/india/healthca…






#SunPharma Dahej unit observations accessed by CNBC-TV18: Details here 🔎
Read here to know more👇
cnbctv18.com/market/sun-pha…
-Hormaz Fatakia ✒️



Negative News for Laurus Labs
#LaurusLabs gets 5 observations grom USFDA Inspection.
#stocktowatch

Strides Pharma Science soars as its step-down arm gets USFDA’s approval for Sucralfate Oral Suspension
investmentguruindia.com/newsdetail/str…
#StockMarket #PharmaSector Strides Pharma Science Limited #USFDA #Investmentguruindia



●Plerixafor Injection. It's intended for patients with non-Hodgkin's lymphoma or multiple myeloma
Gland Pharma has received approval from the United States Food and Drug Administration (USFDA) for plerixafor injection single-dose vial product.
#Pharmanews